© 2003 Lippincott Williams & Wilkins, Inc. Oswestry Disability Index scores and the Physical Component Scores and Pain Index of the SF-36 scale at 3, 6, 12, and 24 months showed statistically superior outcomes in the rhBMP-2 group. The patients treated with rhBMP-2 had statistically superior outcomes with regard to length of surgery, blood loss, hospital stay, reoperation rate, median time to return to work, and fusion rates at 6, 12, and 24 months. Of these patients, 277 had their cages implanted with rhBMP-2 on an absorbable collagen sponge and 402 received autograft transferred from the iliac crest. An integrated analysis of multiple clinical studies was performed using an analysis of covariance to adjust for preoperative variables in a total of 679 patients. While it’s not illegal for a doctor to use a product for off-brand uses, it is for a manufacturer to market its products as such.Multicenter human clinical studies of patients undergoing anterior lumbar fusion have been conducted using recombinant bone morphogenetic protein or rhBMP-2 on an absorbable collagen sponge, marketed as INFUSE Bone Graft, or autograft implanted in the LT-CAGE Lumbar Tapered Fusion device. The InFUSE Bone Graft component induces new bone tissue at the site of implantation. Lawyers have primarily focused on off-label uses with lawsuits. InFUSE Bone Graft consists of recombinant human Bone Morphogenetic Protein-2 (rhBMP-2, known as dibotermin alfa) p laced on an absorbable collagen sponge (ACS). 
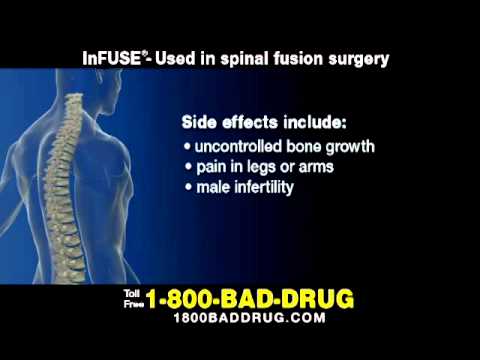
Even before the Yale University study results were published, recipients and employees already filed lawsuits concerning incentives paid to doctors, wrongful death, emergency surgery, fraudulent promotion, and off-label uses. LawsuitsĪccording to statements from Medtronic, more than 1 million patients have received an Infuse Bone Graft, either for standard or off-label treatments. As well, the article found early published data underreported side effects, instead emphasizing favorable results. The product, according to the findings, is associated with a minimal but still increased risk of cancer and may be of little to no benefit compared with traditional treatments. The results same out in June 2013 in Annals of Internal Medicine, revealing Medtronic’s Infuse works as well as a traditional bone graft but may not be safe. The Spine Journal, in July 2011, issued a series of articles about risks associated with the Infuse Bone Graft: Medtronic’s reticence to report the product’s effects from its clinical trials and lack of disclosure about the authors’ financial ties to the company how the Infuse Bone Graft can cause nerve and spinal cord problems and the side effects associated with off-label usage.įollowing these reports, Medtronic announced Yale University would be conducting an independent study on the Infuse Bone Graft. The investigation involved determining whether the surgeons did not report complications, including swelling in neck and throat, abnormal bone growth, and sterility. Senate Finance Committee began investigating the surgeons Medtronic allegedly paid. Department of Justice began looking into the product’s off-label uses, as 280 reports of side effects had been filed with the FDA.īy 2011, the U.S. Patients who suffer from degenerative disk disease, a painful back condition in which discs in the back gradually become more and more. Other reports around this time revealed Medtronic may have paid doctors involved in studies to deliver positive results.Īt the same time, the U.S. Particularly, use of the bone graft in the cervical spine poses risks, has not proven to be effective, and was not FDA-approved. In July 2008, the FDA put out a warning regarding off-label uses for Infuse Bone Grafts. 
Recipients have required respiratory support, a feeding tube, anti-inflammatory medications, tracheotomies, or additional surgery.Īlthough the product stimulates bone growth to replace damaged spinal discs and originally was thought to be an improvement over standard grafts from the hip, Medtronic allegedly marketed the product for off-label procedures, including for cervical fusion, thoracic fusion, posterior fusion, and multi-level fusions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
